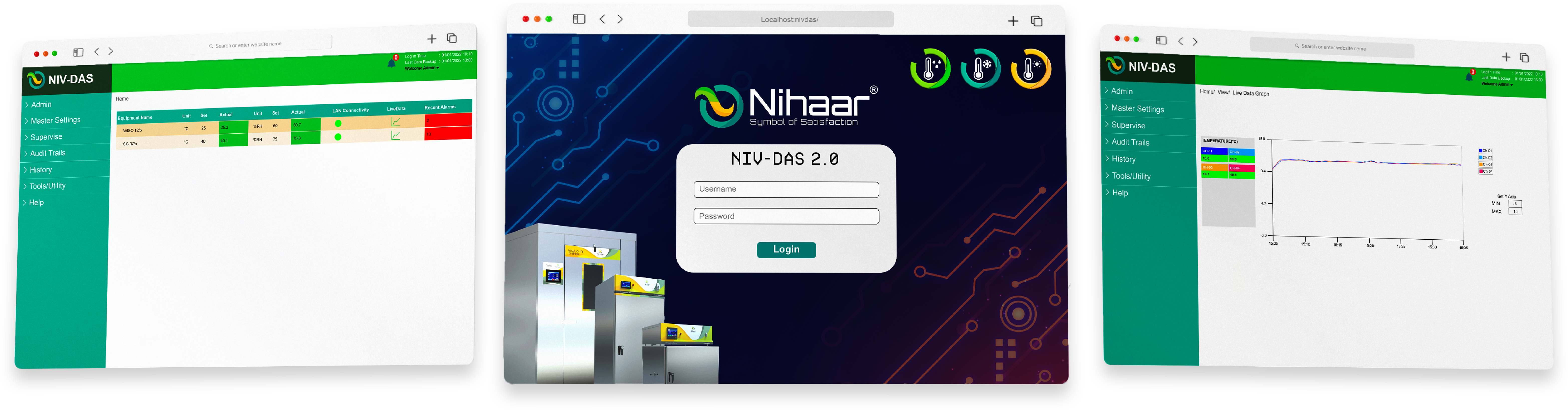
Web-based access.
Connect 16 equipment.
E-signature.
Complete Audit traceability.
Multi‑channel alerts.
Real‑time Data Graph.
Custom MKT calculations.
Auto data backups.
installations
Customers
Years
Size Chamber Supplied
At Nihaar Equipment, every chamber is CE Certified and designed to meet stringent global standards, including ICH Guidelines, WHO, EU-GMP, and US-FDA requirements. With Nihaar, you don't just get precision equipment — you gain confidence in regulatory-ready performance trusted by pharmaceutical leaders worldwide.

Document Holder

Cooling & Humidification

30 Days Data Storage

Door Access System
NIV-DAS is a US-FDA 21 CFR Part 11 compliant monitoring software built to keep your facility audit-ready at all times. With secure electronic records, audit trails, and user authentication, it ensures every action is traceable and compliant with global regulatory standards. NIV-DAS helps pharmaceutical and laboratory teams maintain data integrity, transparency, and effortless compliance — so you're always prepared for inspections with confidence.
Web-based access.
Connect 16 equipment.
E-signature.
Complete Audit traceability.
Multi‑channel alerts.
Real‑time Data Graph.
Custom MKT calculations.
Auto data backups.
Built to meet Global Pharma standards
1Tailor-made chambers for your unique requirements
2AMC, Upgradations and lifelong support
3Trusted by the top pharmaceutical companies worldwide
4